|
These mechanisms include both high temperature and low temperature reaction pathways. 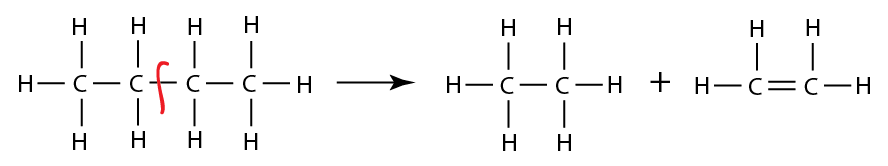  
Explicitly accounting for the surface tension difference between the real and model substances, or alternatively using the Hale scaling parameter, Ω, consistent with the model potential, increases the offset to ~6 orders of = ). For all three alkanes, the experimental and simulated nucleation rates are offset by ~3 orders of magnitude when plotted as a function of ln S/(T c/T – 1) 1.5. Complementary Monte Carlo simulations, using the transferable potentials for phase equilibria-united atom potentials, suggest that despite the high degree of supercooling, the critical clusters remain liquid like under experimental conditions for n-pentane through n-heptane, but adopt more ordered structures for n-octane and n-nonane. 
For n-pentane and n-hexane, these are the first nucleation rate measurements that have been made, and the trends in the current data agree well with those in the earlier work of Ghosh et al. We measured the nucleation rates of n-pentane through n-heptane in a supersonic nozzle at temperatures ranging from ca.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
